ExoQuick PLUS Exosome Purification Kit - for serum and plasma
| Specifications | |
|---|---|
| Product Category: | Exosome Purification |
| Sample Type: | Plasma/Serum |
Product Description
ExoQuick PLUS - Quick and Efficient Exosome Isolation for Sensitive Applications, when you need an extra level of confidence
- Better—delivers as much as 150-fold higher yields than ultracentrifugation-based methods
- Cleaner—dramatically reduces the amount of carry-over protein compared to other column- or precipitation-based kits, and even ultracentrifugation-based methods
- Powerful—detects biomarkers with as much as a 3-fold increase in sensitivity
- Fast—takes less than 10 minutes of hands-on-time after initial ExoQuick isolation
While the ExoQuick® family of reagents speed-up and simplify exosome isolation and are compatible with most downstream uses, some applications are especially sensitive to carry-over of non-exosomal proteins applications such as mass spectrometry, western blotting of low abundance markers, exosome labeling, and in vivo/ex vivo exosome delivery. Which is why SBI offers ExoQuick PLUS and ExoQuick-TC PLUS.
ExoQuick can be used to purify exosomes from plasma, serum, and malignant ascites. With a simple workflow involving minimal hands-on time and low input sample volume requirements, ExoQuick PLUS is an excellent option for researchers who need to purify multiple exosome samples and/or samples from small animal models or clinical research samples that will then be used in sensitive downstream applications.
To isolate exosomes from plasma, we recommend pre-treatment with the Thrombin Plasma Prep or using the ExoQuick™ Plasma Prep and Exosome Precipitation Kit.
How it Works
ExoQuick PLUS further reduces protein carryover from serum/plasma and tissue culture exosomal preparations with a simple treatment by ready-to-use, customized microspheres (kits contains enough ExoQuick and pre-packed microspheres for 10 reactions. In less than 10 minutes of total hands-on time after ExoQuick isolation, you can obtain highly pure, intact exosomes suitable for a variety of protein-sensitive applications.
To isolate exosomes from cleared serum, plasma, or ascites fluid, simply:
- Add an appropriate volume of ExoQuick PLUS to as little as 100 µl sample
- Incubate for at least one hour at 4°C
- Pellet exosomes with a 30-minute low-speed spin (1500g)
- Resuspend pellet, add ExoQuick PLUS Microspheres, and incubate for 10 minutes at 4°C
- Remove microspheres with a 30-minute low-speed spin (1500g)
Exosome Isolation with ExoQuick PLUS saves time and results in higher protein yields and less albumin carryover compared to Ultracentrifugation:
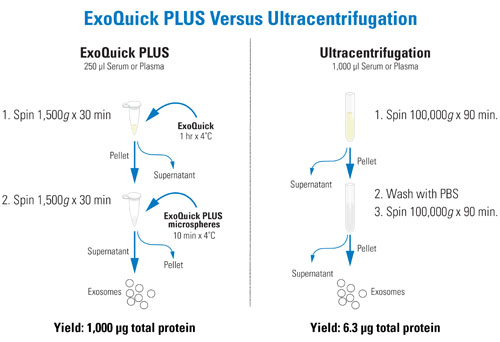
Fig.1. Workflow of ExoQuick PLUS compared to Ultracentrifugation.
Performance Data
ExoQuick PLUS provides efficient exosome isolation with reduced protein carry-over, resulting in isolated exosomes suitable for most sensitive downstream applications.
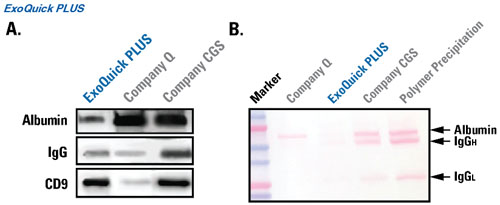
Fig.2 ExoQuick PLUS exosome isolation results in samples with less protein carry-over than competitors’ products. For each lane, 2.5 µg of total protein was loaded. A. Western blot probing for serum albumin, IgG, and the exosome-specific tetraspanin marker CD9 shows higher CD9:IgG and CD9:albumin ratios in the ExoQuick PLUS lane than in the competitors’ lanes. B. Ponceau staining to assess total serum protein abundance shows the least amount of carry-over protein in the ExoQuick PLUS lane.

Fig.3. ExoQuick PLUS exosome isolation results in samples with less protein carry-over than ultracentrifugation. Western blot analysis of EVs isolated using either ExoQuick PLUS or ultracentrifugation and wash demonstrate that the ExoQuick PLUS method results in less co-purification of albumin and IgG for much cleaner EV preps (each lane loaded with 2.5 µg protein equivalent). In addition, the 3-fold higher EV-specific CD9 signal indicates a higher EV yield in the ExoQuick PLUS lane.
More data of performance can be found in the ExoQuick PLUS Product Sheet.
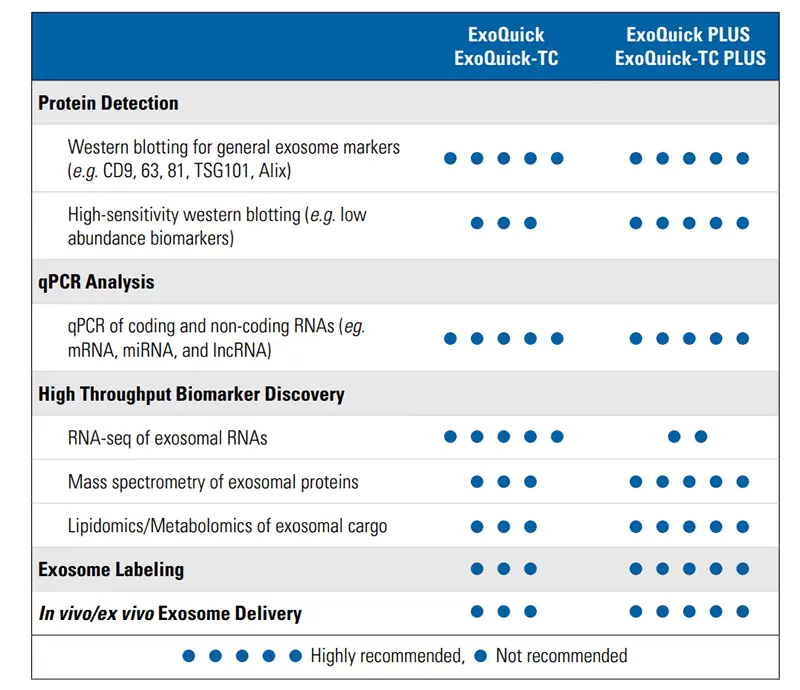
Choose the best ExoQuick product for your application.
- Catalog Number
EQPL10A-1-SBI - Supplier
SBI System Biosciences - Size
- Shipping
Blue Ice

