Plasma/Serum Circulating and Exosomal RNA Purification 96-well Kit (Slurry Format), 0.25 - 2 ml sample input
| Specifications | |
|---|---|
| Product Category: | Circulating & Exosomal RNA incl. microRNA Purification |
| Sample Type: | Plasma, Serum |
Product Description
- Isolate all sizes of circulating RNA
- Process 96 samples in less than 1 hour
- Concentrate circulating RNA into small elution volumes
- Isolate inhibitor-free circulating RNA
Norgen’s Plasma/Serum Circulating RNA Purification 96-Well Kit (Slurry Format) provides a fast, reliable and simple procedure for isolating circulating RNA from plasma and serum samples ranging from 0.25 ml to 2 ml. Free-circulating plasma and serum RNA can serve as both tumor- and fetal-specific markers for cancer detection and prenatal diagnosis. As well, free-circulating RNAs have the potential to provide biomarkers for other disease states. Free-circulating RNA in plasma or serum are usually present as short fragments of less than 1000nt, and free-circulating miRNA (21nt) can also be found in plasma and serum. This kit is able to isolate all sizes of circulating RNA, including microRNA, without the use of phenol or chloroform. The slurry format provides an advantage over other available kits in that it does not require extension tubes for the purification of free-circulating RNA from large sample volumes. RNA can be isolated from either fresh or frozen samples using this kit. The kit allows for the concentration of RNA that is present in low concentrations (1-100ng/ml circulating RNA in human plasma).
Purification is based on using Norgen's proprietary resin as the separation matrix. For the isolation of circulating RNA the plasma/serum samples are first mixed with PS Solution A, which contains the separation matrix, and PS Solution B. Next, ethanol and PS Solution C are added and the slurry is loaded onto a provided 96-Well Filter Plate. This is followed by washing of the bound RNA to remove the remaining proteins and other impurities. Lastly, the purified circulating RNA is eluted into 100 µl of the provided Elution Buffer. The procedure can be performed using either a vacuum manifold or centrifugation. Please see the procedure flowchart to the right.
Preparation time for 96 samples is approximately 1 hour. The purified plasma/serum free-circulating RNA is eluted in an elution solution that is compatible with real time PCR, reverse transcription PCR, Northern blotting, RNase protection and primer extension, and expression array assays.
Performance Data
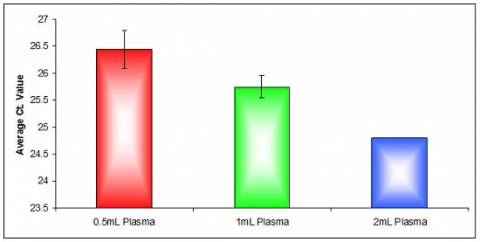
Figure 2. Effective Isolation of Plasma Circulating RNA from Different Volumes. Norgen's Plasma/Serum Circulating RNA Purification Mini Kit (Slurry Format) was used to isolate circulating RNA from 0.5mL, 1mL and 2mL plasma. Three microlitres of the purified RNA was then used as the template in RT-qPCR reactions to detect the human 5S gene. The 5S housekeeping gene was detected from all plasma sample volumes used. The amplification of the 5S rRNA showed an increasing amount of RNA with increasing the sample input volume. This is represented by the decrease of the Ct value with increasing the sample input volume. Average Ct. values for the amplification of the 5S rRNA isolated from 0.5mL plasma is represented by the Red bar, Average Ct. values for the amplification of the 5S rRNA isolated from 1mL plasma is represented by the Green bar whereas Average Ct. values for the amplification of the 5S rRNA isolated from 2mL plasma is represented by the blue bar.
Product Citations
- Arianna Rocca, Francesco Martelli, Serena Delbue, Pasquale Ferrante, Dario Bartolozzi, Alberta Azzi, Simone Giannecchini (2015) The JCPYV DNA load inversely correlates with the viral microrna expression in blood and cerebrospinal fluid of patients at risk of PML Journal of Clinical Virology
- Judie A. Howrylak, Melody Pham, Christiana Oji-Mmuo, Susan DiAngelo, Debra Spear, Kimberly Group, Roopa Siddaiah, Neal Thomas, Patricia Silveyra (2015) Expression Profiling Of MiRNAs In Pediatric Bronchopulmonary Dysplasia American Thoracic Society International Conference Abstracts
- Natasa Zarovni, Antonietta Corrado, Paolo Guazzi, Davide Zocco, Elisa Lari, Giorgia Radano, Jekatarina Muhhina, Costanza Fondelli, Julia Gavrilova, Antonio Chiesi (2015) Integrated isolation and quantitative analysis of exosome shuttled proteins and nucleic acids using immunocapture approaches Methods
- Anna Brunet-Vega, Carles Pericay, María Elisa Quílez, María José Ramírez-Lázaro, Xavier Calvet, Sergio Lario (2015) Variability in microRNA recovery from plasma: comparison of five commercial kits Analytical Biochemistry
- Jagotamoy Das, Ivaylo Ivanov, Laura Montermini, Janusz Rak, Edward H. Sargent & Shana O. Kelley (2015) An electrochemical clamp assay for direct, rapid analysis of circulating nucleic acids in serum Nature Chemistry
- Catalog Number
29500-NB - Supplier
Norgen Biotek - Size
- Shipping
RT

