Lentiviral Packaging Service from SBI
Accelerate your Research with SBI's Lentiviral Technology and Packaging Expertise
Recombinant lentiviral vectors have been shown to be powerful tools for stable gene transfer to both dividing and non-dividing cells in vitro and in vivo.
- Obtain ready-to-transduce, packaged pseudoviral particles
- Delivery into a wide range of cell lines
- 3 different levels of titer available to meet your specific requirements
- Avoid any biosafety concerns to package virus in your lab with 3rd generation biosafe lentiviral particles
- SBI´s scientists routinely prepare viral concentrations >10^9 IFUs / mL
- Ultra-high titer virus available - perfect for in vivo and stem cell applications
- Concentrated viral preparations without ultracentrifugation means less toxic debris
- Titers are measured using the UltraRapid Titer Kit - real infectious titer is measured, not an envelope protein
Through years of experience with lentiviral vectors, our partner SBI developed their own proprietary packaging systems and efficient protocols for the rapid generation of pseudoviral particles. Because all aspects of this service are completed on-site in SBI‘s Palo Alto, CA, facility, we can offer complete confidentiality, transparency on project status, and fully-informed, expert technical support.
You can order lentiviral packaging services using lentivectors provided by you as well as lentivectors constructed by SBI to obtain ready-to-use particles
Available Titer Levels
|
TITER |
IFU/ML |
APPLICATION |
|
Regular Titer |
>107 |
Standard cell culture models |
|
High Titer |
>108 |
More difficult-to-transduce cells (e.g. suspension cells) |
|
Ultra-high Titer |
>109 |
Very difficult-to-transduce cells such as stem cells and primary cells; In vivo applications |
When providing a construct for a lentiviral packaging service, please fill out the Lentiviral Plasmid Information Sheet and send it to info@biocat.com - when you are interested in the packaging of a premade SBI construct, please let us know which one you like to be packaged and ask for a quotation, as discounts might apply.
Titers are guaranteed, when the construct to be packaged is based on an SBI vector. If it is not an SBI vector construct, please let us know the vector details and we´ll check if a titer guarantee can be given.
Supporting Data
Lentivirus produced by SBI exhibits broad tropism and robust in vivo transduction efficiency.
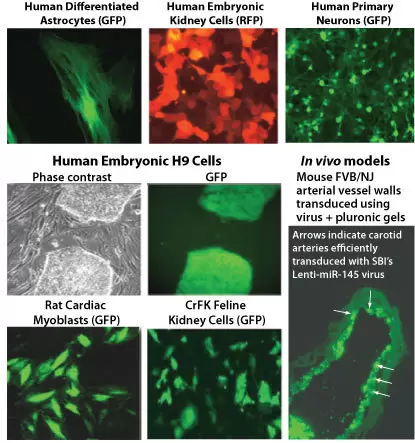
Fig.1. SBI’s packaged lentivirus exhibits broad tropism.
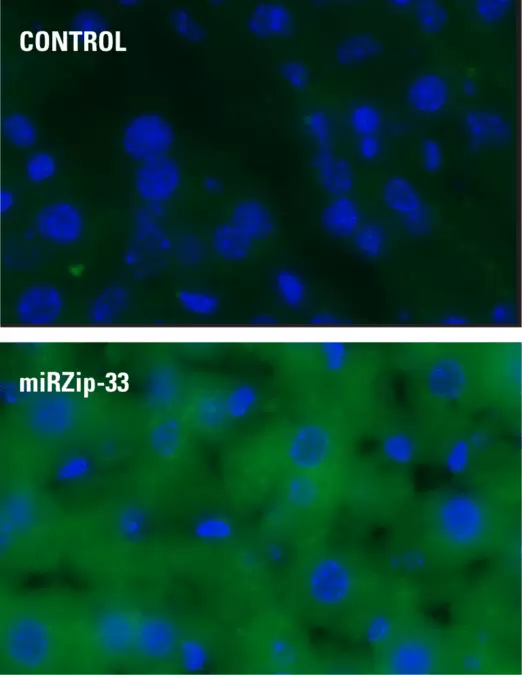
Fig.2. Lentivirus produced by SBI delivers high transduction efficiency in vivo. DAPI and GFP imaging of mouse liver six days after infection with either no virus (top) or miRZip-33 virus with a GFP marker (bottom). Lentivirus was introduced into mice via retroorbital injection at 2 x 109 IFU/mouse. Rayner, KJ, et al. miR-33 Contributes to the Regulation of Cholesterol Homeostasis. Science. 2010 June 18. 328(5985):1570. Supporting Online Material. PMCID: PMC3114628.